IUXTA 3D EG®2.0 digital workflow
EAGLEGRID® 2.0 is the digital revision of the previous iuxta-bone implant. Thanks to new technologies, this medical device, already compliant with the new European directive on implantology of 2020, solves all those cases of severe atrophy of the upper jaw or mandible where the residual bone does not allow the use of endosseous implants. Its extreme precision along with the patient’s receiving cortical bone is then improved by stabilization by means of appropriate synthesis screws, which will the EAGLEGRID® 2.0 the certainty of bone integration.
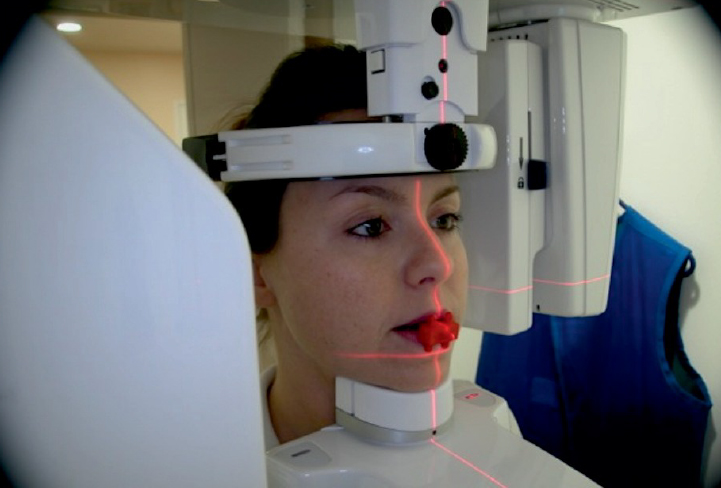
TC Cone Beam and 3D virtual bone model production
The design begins with the acquisition of the patient’s tomographic examination using a Cone Beam. During the examination, the patient must wear an appropriate radiological template that will allow us to design and build a prosthetically guided EAGLEGRID® 2.0.
The acquired DICOM files are sent by the dentist via web to the EagleGrid team who will check its quality and the feasibility of the design.
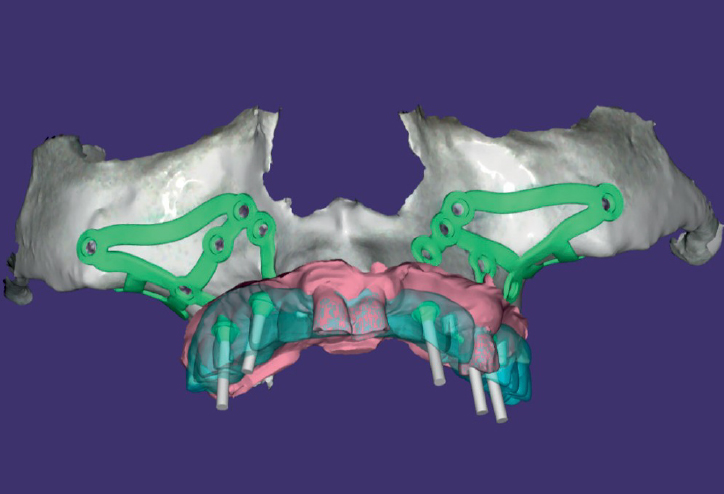
EagleGrid®2.0 digital processing
EAGLEGRID® 2.0 is modelled virtually on the anatomy of the patient by clinical and dental technicians using dedicated software. The design and geometry of the grid is designed to support the correct prosthetical loads ensuring the maximum possible long term success. The entire, virtual design is shared with the dentist in order to make any appropriate changes. After validation by the examining physician, EAGLEGRID® 2.0 is put into production.

SLM (Selective Laser Meldng) 3D printing
EAGLEGRID® 2.0 is produced in Titanium by means of the Laser Melting technique: homogeneous layers of Grade 5 Titanium powders are syntherized by a high intensity Laser beam in gradually successive layers until a 3D object is obtained. The final subperiosteal grid is characterized by a high standard of purity and by a micro-structural homogeneity which ensures high mechanical performance both in the case of static loads and fatigue cycles. At the end of the procedure, each EAGLEGRID® 2.0 is laser-etched with an alphanumeric code which uniquely matches it to the patient for whom it is intended.

Cleaning, decontamination, packaging and shipping
EAGLEGRID®2.0 are cleaned and decontaminated in automatic ultrasound machines in a special structure called a clean room, in a controlled environment. After packaging, they are gamma-ray sterilized. All EAGLEGRID® 2.0 production cycles are checked and recorded in order to guarantee product traceability in compliance with the most advanced and restrictive reference standards.
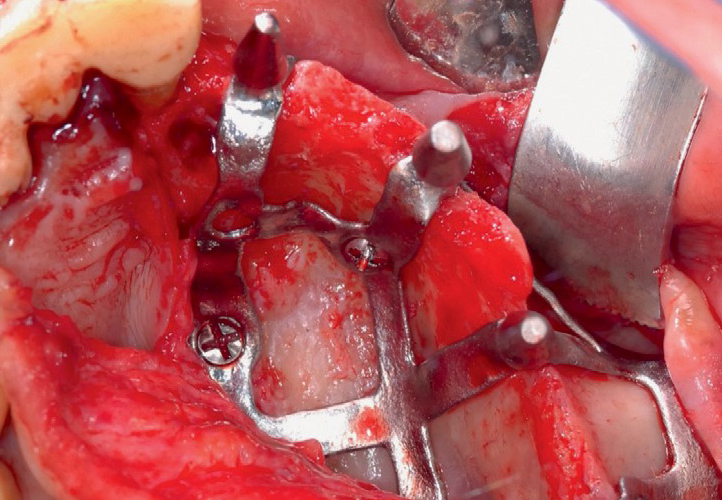
Surgery
Surgery takes place under local anesthesia or with conscious sedation. EAGLEGRID Polska can provide the operator with a whole series of aids to facilitate the positioning of the grid, not least the supply of a first temporary milled PMMA bridge that will allow the patient to chew immediately, pending perfect healing of the underlying soft tissues.